Decontamination of Arsenic in Actual Water Samples by Calcium Containing Layered Double Hydroxides from a Convenient Synthesis Method
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
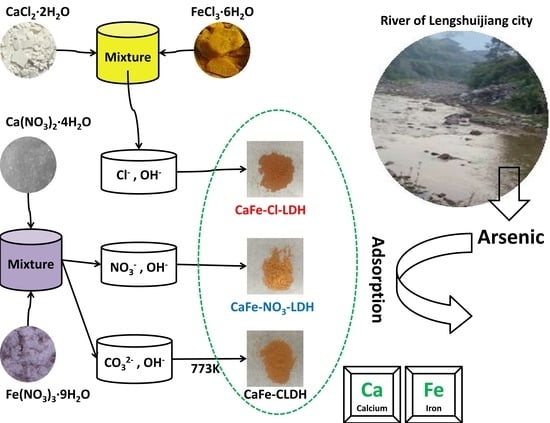
2.2. Synthesis of Materials
2.3. Characterization and Analysis
2.4. Batch Adsorption Experiments
3. Results and Discussion
3.1. Characterization
3.1.1. X-ray Diffraction (XRD)
3.1.2. FTIR Analysis
3.1.3. Analysis of Specific Surface Area and Pore Size Distribution
3.1.4. Morphological Analysis
3.2. Adsorption of Arsenic
3.2.1. Isotherms of Arsenic Adsorption
3.2.2. Kinetics of Arsenic Adsorption
3.2.3. Effect of pH on Arsenic Adsorption
3.2.4. Effect of Materials Dosage on Arsenic Adsorption
3.2.5. Effects of Anions on Arsenic Adsorption
3.2.6. Adsorption and Removal of Arsenic in Actual Water Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ravenscroft, P.; Brammer, H.; Richards, K. Arsenic Pollution: A Global Synthesis; Wiley-Blackwell: Oxford, UK, 2009; p. 588. [Google Scholar]
- Rodriguez-Lado, L.; Sun, G.; Berg, M.; Zhang, Q.; Xue, H.; Zheng, Q.; Johnson, C.A. Groundwater arsenic contamination throughout china. Science 2013, 341, 866–868. [Google Scholar] [CrossRef] [PubMed]
- Mandal, B.K.; Suzuki, K.T. Arsenic round the world: A review. Talanta 2002, 58, 201–235. [Google Scholar] [CrossRef]
- Alonso, D.L.; Latorre, S.; Castillo, E.; Brandao, P.F.B. Environmental occurrence of arsenic in colombia: A review. Environ. Pollut. 2014, 186, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Bundschuh, J.; Litter, M.I.; Parvez, F.; Roman-Ross, G.; Nicolli, H.B.; Jean, J.S.; Liu, C.W.; Lopez, D.; Armienta, M.A.; Guilherme, L.R.G.; et al. One century of arsenic exposure in latinamerica: A review of history and occurrence from 14 countries. Sci. Total Environ. 2012, 429, 2–35. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.L.; Yang, T.; Chen, Q.Y.; Liu, F.; Wang, B.B. Distribution and potential ecological risk of heavy metals in the typical eco-units of haihe river basin. Front. Environ. Sci. Eng. 2016, 10, 103–113. [Google Scholar] [CrossRef]
- Meng, Z.G.; Korfiatis, G.P.; Christodoulatos, C.; Bang, S. Treatment of arsenic in bangladesh well water using a household co-precipitation and filtration system. Water Res. 2001, 35, 2805–2810. [Google Scholar] [CrossRef]
- Śmiech, K.; Tolsma, A.; Kovács, T.; Dalbosco, V.; Yasadi, K.; Groendijk, L.; Agostinho, L. Comparing mixed-media and conventional slow-sand filters for arsenic removal from groundwater. Water 2018, 10, 119. [Google Scholar] [CrossRef]
- Rahman, M.A.; Hasegawa, H.; Ueda, K.; Maki, T.; Okumura, C.; Rahman, M.M. Arsenic accumulation in duckweed (Spirodelapolyrhiza L.): A good option for phytoremediation. Chemosphere 2007, 69, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.S.; Xu, W.Z.; Shen, H.L.; Yan, H.L.; Xu, W.X.; He, Z.Y.; Ma, M. Engineering arsenic tolerance and hyperaccumulation in plants for phytoremediation by a PvACR3 transgenic approach. Environ. Sci. Technol. 2013, 47, 9355–9362. [Google Scholar] [CrossRef] [PubMed]
- Van Genuchten, C.M.; Addy, S.E.A.; Pena, J.; Gadgil, A.J. Removing arsenic from synthetic groundwater with iron electrocoagulation: An Fe and as K-edge EXAFS study. Environ. Sci. Technol. 2012, 46, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.K.; Zhou, G.M.; Xiong, X.M.; Guan, X.H.; Li, L.N.; Bao, H.L. Enhanced arsenite removal from water by Ti(SO4)2 coagulation. Water Res. 2013, 47, 4340–4348. [Google Scholar] [CrossRef] [PubMed]
- Nidheesh, P.V.; Singh, T.S.A. Arsenic removal by electrocoagulation process: Recent trends and removal mechanism. Chemosphere 2017, 181, 418–432. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Kang, M.; Kamei, T.; Magara, Y. Performance of nanofiltration for arsenic removal. Water Res. 2002, 36, 3371–3377. [Google Scholar] [CrossRef]
- Uddin, M.T.; Mozumder, M.S.I.; Islam, M.A.; Deowan, S.A.; Hoinkis, J. Nanofiltration membrane process for the removal of arsenic from drinking water. Chem. Eng. Technol. 2007, 30, 1248–1254. [Google Scholar] [CrossRef]
- Awual, M.R.; Hossain, M.A.; Shenashen, M.A.; Yaita, T.; Suzuki, S.; Jyo, A. Evaluating of arsenic(V) removal from water by weak-base anion exchange adsorbents. Environ. Sci. Pollut. Res. 2013, 20, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Anirudhan, T.S.; Unnithan, M.R. Arsenic(V) removal from aqueous solutions using an anion exchanger derived from coconut coir pith and its recovery. Chemosphere 2007, 66, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Qi, P.; Pichler, T. Competitive adsorption of As (III) and As (V) by ferrihydrite: Equilibrium, kinetics, and surface complexation. Water Air Soil Pollut. 2016, 227, 387. [Google Scholar] [CrossRef]
- Ma, J.; Guo, H.; Lei, M.; Zhou, X.; Li, F.; Yu, T.; Wei, R.; Zhang, H.; Zhang, X.; Wu, Y. Arsenic adsorption and its fractions on aquifer sediment: Effect of pH, arsenic species, and iron/manganese minerals. Water Air Soil Pollut. 2015, 226, 260. [Google Scholar] [CrossRef]
- Usman, M.; Katsoyiannis, I.; Mitrakas, M.; Zouboulis, A.; Ernst, M. Performance evaluation of small sized powdered ferric hydroxide as arsenic adsorbent. Water 2018, 10, 957. [Google Scholar] [CrossRef]
- Singh, R.; Singh, S.; Parihar, P.; Singh, V.P.; Prasad, S.M. Arsenic contamination, consequences and remediation techniques: A review. Ecotoxicol. Environ. Saf. 2015, 112, 247–270. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Paul, B. The global menace of arsenic and its conventional remediation—A critical review. Chemosphere 2016, 158, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.-H.; Lim, T.-T.; Dong, Z. Application of layered double hydroxides for removal of oxyanions: A review. Water Res. 2008, 42, 1343–1368. [Google Scholar] [CrossRef] [PubMed]
- Cavani, F.; Trifirò, F.; Vaccari, A. Hydrotalcite-type anionic clays: Preparation, properties and applications. Catal. Today 1991, 11, 173–301. [Google Scholar] [CrossRef]
- Zubair, M.; Daud, M.; McKay, G.; Shehzad, F.; Al-Harthi, M.A. Recent progress in layered double hydroxides (LDH)-containing hybrids as adsorbents for water remediation. Appl. Clay Sci. 2017, 143, 279–292. [Google Scholar] [CrossRef]
- Asiabi, H.; Yamini, Y.; Shamsayei, M. Highly selective and efficient removal of arsenic(V), chromium(VI) and selenium(VI) oxyanions by layered double hydroxide intercalated with zwitterionic glycine. J. Hazard. Mater. 2017, 339, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.J.; Yu, X.L.; Tong, S.R.; Ge, M.F.; Zuo, J.C.; Cao, C.Y.; Song, W.G. Performance and mechanism of Mg/Fe layered double hydroxides for fluoride and arsenate removal from aqueous solution. Chem. Eng. J. 2013, 228, 731–740. [Google Scholar] [CrossRef]
- Al-Jaberi, M.; Naille, S.; Dossot, M.; Ruby, C. Inter layer interaction in Ca-Fe layered double hydroxides intercalated with nitrate and chloride species. J. Mol. Struct. 2015, 1102, 253–260. [Google Scholar] [CrossRef]
- Sipiczki, M.; Srankó, D.F.; Kukovecz, Á.; Kónya, Z.; Sipos, P.; Pálinkó, I. Synthesis and characterisation of alkaline earth-iron(III) double hydroxides. Chem. Pap. 2011, 65, 840–846. [Google Scholar] [CrossRef]
- Ferencz, Z.; Szabados, M.; Varga, G.; Csendes, Z.; Kukovecz, A.; Konya, Z.; Carlson, S.; Sipos, P.; Palinko, I. Mechanochemical synthesis and intercalation of Ca(II)Fe(III)-layered double hydroxides. J. Solid State Chem. 2016, 233, 236–243. [Google Scholar] [CrossRef]
- Sun, Z.; Park, J.-S.; Kim, D.; Shin, C.-H.; Zhang, W.; Wang, R.; Rao, P. Synthesis and adsorption properties of ca-al layered double hydroxides for the removal of aqueous fluoride. Water Air Soil Pollut. 2017, 228, 23. [Google Scholar] [CrossRef]
- Abdellattif, M.; Mokhtar, M. MgAl-layered double hydroxide solid base catalysts for henry reaction: A green protocol. Catalysts 2018, 8, 133. [Google Scholar] [CrossRef]
- Sipiczki, M.; Kuzmann, E.; Homonnay, Z.; Megyeri, J.; Palinko, I.; Sipos, P. The structure and stability of cafe layered double hydroxides with various Ca:Fe ratios studied by mossbauer spectroscopy, X-ray diffractometry and microscopic analysis. J. Mol. Struct. 2013, 1044, 116–120. [Google Scholar] [CrossRef]
- Gong, Z.; Lu, X.; Ma, M.; Watt, C.; Le, X.C. Arsenic speciation analysis. Talanta 2002, 58, 77–96. [Google Scholar] [CrossRef]
- Rahman, M.T.; Kameda, T.; Kumagai, S.; Yoshioka, T. Adsorption isotherms and kinetics of arsenic removal from aqueous solution by mg-al layered double hydroxide intercalated with nitrate ions. React. Kinet. Mech. Catal. 2017, 120, 703–714. [Google Scholar] [CrossRef]
- Ma, L.J.; Islam, S.M.; Liu, H.Y.; Zhao, J.; Sun, G.B.; Li, H.F.; Ma, S.L.; Kanatzidis, M.G. Selective and efficient removal of toxic oxoanions of As (III), As (V), and Cr (VI) by layered double hydroxide intercalated with MoS42−. Chem. Mater. 2017, 29, 3274–3284. [Google Scholar] [CrossRef]
- Miller, F.A.; Wilkins, C.H. Infrared spectra and characteristic frequencies of inorganic ions. Anal. Chem. 1952, 24, 1253–1294. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared and raman spectra of inorganic and coordination compounds. In Handbook of Vibrational Spectroscopy; John Wiley & Sons, Ltd.: London, UK, 2006. [Google Scholar]
- Balbuena, P.B.; Gubbins, K.E. Classification of adsorption behavior: Simple fluids in pores of slit-shaped geometry. Fluid Phase Equilib. 1992, 76, 21–35. [Google Scholar] [CrossRef]
- Carja, G.; Nakamura, R.; Aida, T.; Niiyama, H. Textural properties of layered double hydroxides: Effect of magnesium substitution by copper or iron. Microporous Mesoporous Mater. 2001, 47, 275–284. [Google Scholar] [CrossRef]
- Guo, Y.W.; Zhu, Z.L.; Qiu, Y.L.; Zhao, J.F. Adsorption of arsenate on Cu/Mg/Fe/La layered double hydroxide from aqueous solutions. J. Hazard. Mater. 2012, 239, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. II. Liquids. J. Am. Chem. Soc. 1917, 39, 1848–1906. [Google Scholar] [CrossRef]
- Freundlich, H. Colloid and Capillary Chemistry; Methuen: London, UK, 1926. [Google Scholar]
- Zhang, S.; Niu, H.; Cai, Y.; Zhao, X.; Shi, Y. Arsenite and arsenate adsorption on coprecipitated bimetal oxide magnetic nanomaterials: MnFe2O4 and CoFe2O4. Chem. Eng. J. 2010, 158, 599–607. [Google Scholar] [CrossRef]
- Chammui, Y.; Sooksamiti, P.; Naksata, W.; Thiansem, S.; Arqueropanyo, O.A. Removal of arsenic from aqueous solution by adsorption on leonardite. Chem. Eng. J. 2014, 240, 202–210. [Google Scholar] [CrossRef]
- Turk, T. Removal of dissolved arsenic by pyrite Ash waste. Mine Water Environ. 2017, 36, 255–263. [Google Scholar] [CrossRef]
- Chen, W.F.; Parette, R.; Zou, J.Y.; Cannon, F.S.; Dempsey, B.A. Arsenic removal by iron-modified activated carbon. Water Res. 2007, 41, 1851–1858. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.H.; Wang, J.; Wang, L.; Sheng, G.P.; Liu, J.H.; Yu, H.Q.; Huang, X.J. Enhanced arsenic removal from water by hierarchically porous CeO2-ZrO2nanospheres: Role of surface- and structure-dependent properties. J. Hazard. Mater. 2013, 260, 498–507. [Google Scholar] [CrossRef] [PubMed]
- Violante, A.; Pucci, M.; Cozzolino, V.; Zhu, J.; Pigna, M. Sorption/desorption of arsenate on/from Mg-Al layered double hydroxides: Influence of phosphate. J. Colloid Interface Sci. 2009, 333, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Ardau, C.; Frau, F.; Lattanzi, P. New data on arsenic sorption properties of Zn-Al sulphate layered double hydroxides: Influence of competition with other anions. Appl. Clay Sci. 2013, 80–81, 1–9. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water Res. 2000, 34, 735–742. [Google Scholar] [CrossRef]
- Bektas, N.; Aydin, S.; Oncel, M.S. The adsorption of arsenic ions using beidellite, zeolite, and sepiolite clays: A study of kinetic, equilibrium and thermodynamics. Sep. Sci. Technol. 2011, 46, 1005–1016. [Google Scholar] [CrossRef]
- Taty-Costodes, V.C.; Fauduet, H.; Porte, C.; Delacroix, A. Removal of Cd(II) and Pb(II) ions, from aqueous solutions, by adsorption onto sawdust of Pinussylvestris. J. Hazard. Mater. 2003, 105, 121–142. [Google Scholar] [CrossRef] [PubMed]
- Giménez, J.; Martínez, M.; de Pablo, J.; Rovira, M.; Duro, L. Arsenic sorption onto natural hematite, magnetite, and goethite. J. Hazard. Mater. 2007, 141, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Raven, K.P.; Jain, A.; Loeppert, R.H. Arsenite and arsenate adsorption on ferrihydrite: Kinetics, equilibrium, and adsorption envelopes. Environ. Sci. Technol. 1998, 32, 344–349. [Google Scholar] [CrossRef]
- Chetia, M.; Goswamee, R.L.; Banerjee, S.; Chatterjee, S.; Singh, L.; Srivastava, R.B.; Sarma, H.P. Arsenic removal from water using calcined Mg–Al layered double hydroxide. Clean Technol. Environ. Policy 2012, 14, 21–27. [Google Scholar] [CrossRef]
- Yoshida, M.; Koilraj, P.; Qiu, X.; Hirajima, T.; Sasaki, K. Sorption of arsenate on MgAl and MgFe layered double hydroxides derived from calcined dolomite. J. Environ. Chem. Eng. 2015, 3, 1614–1621. [Google Scholar] [CrossRef]








| Materials | BET Surface Area (m2·g−1) | C-Value in BET Equation | Pore Volume (cm3·g−1) | Average Pore Diameters (nm) |
|---|---|---|---|---|
| CaFe-CLDH | 21.17 | 18.14 | 0.5109 | 3.709 |
| CaFe-Cl-LDH | 20.68 | 70.74 | 0.4122 | 3.135 |
| CaFe-NO3-LDH | 43.31 | 100.3 | 0.3037 | 2.359 |
| Materials | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| Qm (mg·g−1) | KL (L·mg−1) | R2 | KF (mg·g−1) (L·mg−1)−1/n | n | R2 | |
| CaFe-CLDH | 156.0 | 0.05848 | 0.9626 | 24.31 | 2.514 | 0.9920 |
| CaFe-Cl-LDH | 150.5 | 0.03509 | 0.8241 | 19.57 | 2.504 | 0.9424 |
| CaFe-NO3-LDH | 148.0 | 0.04894 | 0.9448 | 17.64 | 2.245 | 0.9524 |
| Adsorbents | Concentration Range (mg·L−1) | pH | Adsorption Capacity (mg·g−1) | Reference |
|---|---|---|---|---|
| Leonardite char | 1–80 | 7.0 | 8.4 | [45] |
| Pyrite Ash | 0.01–0.5 | 7.0 | 0.295 | [46] |
| UltraCarb | 20–22 | 6.0 | 51.3 | [47] |
| CeO2–ZrO2 nanospheres | 0.5–60 | 6.9 | 145.35 | [48] |
| Mg-Fe-Cl-LDH | 3.75–562.5 | 6.0 | 129.5 | [49] |
| Zn-Al-SO4-LDH | 900 | 9.0 | 74.9 | [50] |
| CaFe-CLDH | 1–100 | 6.0 | 156.0 | This study |
| CaFe-Cl-LDH | 1–100 | 6.0 | 150.5 | This study |
| CaFe-NO3-LDH | 1–100 | 6.0 | 148.0 | This study |
| Materials | Pseudo First-Order | Pseudo Second-Order | ||||
|---|---|---|---|---|---|---|
| Qe (mg·g−1) | K1 (min−1) | R2 | Qe (mg·g−1) | K2 (g·mg−1·min−1) | R2 | |
| CaFe-CLDH | 25.89 | 0.0461 | 0.8044 | 27.84 | 0.00226 | 0.9077 |
| CaFe-Cl-LDH | 16.50 | 0.0539 | 0.9378 | 17.30 | 0.00517 | 0.9856 |
| CaFe-NO3-LDH | 16.43 | 0.1054 | 0.9428 | 17.23 | 0.00948 | 0.9832 |
| Sample | As | Ba | Mg | Fe | Ca | K | Cu | Na | Al | P | Sb |
|---|---|---|---|---|---|---|---|---|---|---|---|
| I | 0.03 | 0.03 | 9.24 | 0.54 | 6.89 | 1.9 | 0.01 | 10.62 | 0.47 | 0 | 0.18 |
| II | 1.11 | 0.04 | 7.69 | 0 | 6.64 | 9.8 | 0.01 | 36.09 | 0.21 | 0.02 | 0.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, H.; Liu, S.; Zhang, H.; Qiu, Y.; Zhao, J.; Zhu, Z. Decontamination of Arsenic in Actual Water Samples by Calcium Containing Layered Double Hydroxides from a Convenient Synthesis Method. Water 2018, 10, 1150. https://doi.org/10.3390/w10091150
Lu H, Liu S, Zhang H, Qiu Y, Zhao J, Zhu Z. Decontamination of Arsenic in Actual Water Samples by Calcium Containing Layered Double Hydroxides from a Convenient Synthesis Method. Water. 2018; 10(9):1150. https://doi.org/10.3390/w10091150
Chicago/Turabian StyleLu, Hongtao, Shuxia Liu, Hua Zhang, Yanling Qiu, Jianfu Zhao, and Zhiliang Zhu. 2018. "Decontamination of Arsenic in Actual Water Samples by Calcium Containing Layered Double Hydroxides from a Convenient Synthesis Method" Water 10, no. 9: 1150. https://doi.org/10.3390/w10091150