Abstract
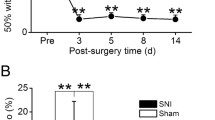
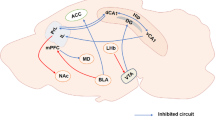
Chronic pain during adolescence can lead to mental health disorders in adulthood, but the underlying mechanism is still unclear. Furthermore, the homeostasis of cerebral glucose metabolism and neurotransmitter metabolic kinetics are closely associated with cognitive development and pain progression. The present study investigated changes in cognitive function and glucose metabolism in adult rats, which had experienced chronic pain during their adolescence. Here, spared nerve injury (SNI) surgery was conducted in 4-week-old male rats. Mechanical nociceptive reflex thresholds were analyzed, and SNI chronic pain (SNI-CP) animals were screened. Based on animal behavioral tests (open field, three-chambered social, novel object recognition and the Y maze), the SNI-CP animals showed learning and memory impairment and anxiety-like behaviors, compared to SNI no chronic pain (SNI-NCP) animals. The cerebral glucose metabolism in the prefrontal cortex and hippocampus of adult SNI-CP animals was decreased with positron emission tomography/computed tomography. GABA2 and Glu4 levels in the metabolic kinetics study were significantly decreased in the hippocampus, frontal cortex, and temporal cortex, and the expression of GLUT3 and GLUT4 was also significantly downregulated in the prefrontal cortex and hippocampus of adult rats in the SNI-CP group. These findings suggest that the rats which suffered chronic pain during adolescence have lower cerebral glucose metabolism in the cortex and hippocampus, which could be related to cognitive function during the development of the central nervous system.







Similar content being viewed by others
Availability of Data and Materials
All raw data and materials during the current study are available from the corresponding author upon reasonable request.
References
Kashyap MP, Roberts C, Waseem M, Tyagi P (2018) Drug targets in neurotrophin signaling in the central and peripheral nervous system. Mol Neurobiol 55(8):6939–6955. https://doi.org/10.1007/s12035-018-0885-3
Shantanu PA, Sharma D, Sharma M, Vaidya S, Sharma K, Kalia K, Tao YX, Shard A et al (2019) Kinesins: motor proteins as novel target for the treatment of chronic pain. Mol Neurobiol 56(6):3854–3864. https://doi.org/10.1007/s12035-018-1327-y
Bonezzi C, Fornasari D, Cricelli C, Magni A, Ventriglia G (2020) Not all pain is created equal: basic definitions and diagnostic work-up. Pain Ther 9(Suppl 1):1–15. https://doi.org/10.1007/s40122-020-00217-w
Descalzi G, Mitsi V, Purushothaman I, Gaspari S, Avrampou K, Loh YE, Shen L, Zachariou V (2017) Neuropathic pain promotes adaptive changes in gene expression in brain networks involved in stress and depression. Sci Signal 10 (471). https://doi.org/10.1126/scisignal.aaj1549
Zhou W, Jin Y, Meng Q, Zhu X, Bai T, Tian Y, Mao Y, Wang L et al (2019) A neural circuit for comorbid depressive symptoms in chronic pain. Nat Neurosci 22(10):1649–1658. https://doi.org/10.1038/s41593-019-0468-2
Husak AJ, Bair MJ (2020) Chronic pain and sleep disturbances: a pragmatic review of their relationships, comorbidities, and treatments. Pain Med (Malden, Mass) 21(6):1142–1152. https://doi.org/10.1093/pm/pnz343
Moriarty O, McGuire BE, Finn DP (2011) The effect of pain on cognitive function: a review of clinical and preclinical research. Prog Neurobiol 93(3):385–404. https://doi.org/10.1016/j.pneurobio.2011.01.002
Tracey I, Mantyh PW (2007) The cerebral signature for pain perception and its modulation. Neuron 55(3):377–391. https://doi.org/10.1016/j.neuron.2007.07.012
Hooten WM (2016) Chronic pain and mental health disorders: shared neural mechanisms, epidemiology, and treatment. Mayo Clin Proc 91 (7):955-970. https://doi.org/10.1016/j.mayocp.2016.04.029 %/ Copyright © 2016 Mayo Foundation for Medical Education and Research. Published by Elsevier Inc. All rights reserved.
Baliki MN, Chialvo DR, Geha PY, Levy RM, Harden RN, Parrish TB, Apkarian AV (2006) Chronic pain and the emotional brain: specific brain activity associated with spontaneous fluctuations of intensity of chronic back pain. J Neurosci 26(47):12165–12173. https://doi.org/10.1523/jneurosci.3576-06.2006
Bushnell MC, Ceko M, Low LA (2013) Cognitive and emotional control of pain and its disruption in chronic pain. Nat Rev Neurosci 14(7):502–511. https://doi.org/10.1038/nrn3516
Corradi-Dell’Acqua C, Tusche A, Vuilleumier P, Singer T (2016) Cross-modal representations of first-hand and vicarious pain, disgust and fairness in insular and cingulate cortex. Nat Commun 7:10904. https://doi.org/10.1038/ncomms10904
Simpson JJ, Drevets WC, Snyder AZ, Gusnard DA, Raichle ME (2001) Emotion-induced changes in human medial prefrontal cortex: II. During anticipatory anxiety. Proc Natl Acad Sci U S A 98(2):688–693. https://doi.org/10.1073/pnas.98.2.688
Liossi C, Howard RF (2016) Pediatric chronic pain: biopsychosocial assessment and formulation. Pediatrics 138 (5). https://doi.org/10.1542/peds.2016-0331 %/ Copyright © 2016 by the American Academy of Pediatrics.
Chehadi O, Rusu AC, Konietzny K, Schulz E, Köster O, Schmidt-Wilcke T, Hasenbring MI (2018) Brain structural alterations associated with dysfunctional cognitive control of pain in patients with low back pain. Eur J Pain (London, England) 22(4):745–755. https://doi.org/10.1002/ejp.1159
Khera T, Rangasamy V (2021) Cognition and pain: a review. Front Psychol 12:673962. https://doi.org/10.3389/fpsyg.2021.673962
Dahlke LA, Sable JJ, Andrasik F (2017) Behavioral therapy: emotion and pain, a common anatomical background. Neurol Sci 38(Suppl 1):157–161. https://doi.org/10.1007/s10072-017-2928-3
Malfliet A, Coppieters I, Van Wilgen P, Kregel J, De Pauw R, Dolphens M, Ickmans K (2017) Brain changes associated with cognitive and emotional factors in chronic pain: a systematic review. Eur J Pain (London, England) 21(5):769–786. https://doi.org/10.1002/ejp.1003
Lau JYF, Heathcote LC, Beale S, Gray S, Jacobs K, Wilkinson N, Crombez G (2018) Cognitive biases in children and adolescents with chronic pain: a review of findings and a call for developmental research. J Pain 19(6):589–598. https://doi.org/10.1016/j.jpain.2018.01.005
You Z, Zhang S, Shen S, Yang J, Ding W, Yang L, Lim G, Doheny JT et al (2018) Cognitive impairment in a rat model of neuropathic pain: role of hippocampal microtubule stability. Pain 159(8):1518–1528. https://doi.org/10.1097/j.pain.0000000000001233
Shiraishi S, Kobayashi H, Nihashi T, Kato K, Iwano S, Nishino M, Ishigaki T, Ikeda M et al (2006) Cerebral glucose metabolism change in patients with complex regional pain syndrome: a PET study. Radiat Med 24(5):335–344. https://doi.org/10.1007/s11604-006-0035-0
Butterfield DA, Halliwell B (2019) Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nat Rev Neurosci 20(3):148–160. https://doi.org/10.1038/s41583-019-0132-6
Hannibal KE, Bishop MD (2014) Chronic stress, cortisol dysfunction, and pain: a psychoneuroendocrine rationale for stress management in pain rehabilitation. Phys Ther 94(12):1816–1825. https://doi.org/10.2522/ptj.20130597
Coppieters I, Ickmans K, Cagnie B, Nijs J, De Pauw R, Noten S, Meeus M (2015) Cognitive performance is related to central sensitization and health-related quality of life in patients with chronic whiplash-associated disorders and fibromyalgia. Pain Physician 18(3):E389-401
Guida F, De Gregorio D, Palazzo E, Ricciardi F, Boccella S, Belardo C, Iannotta M, Infantino R et al (2020) Behavioral, biochemical and electrophysiological changes in spared nerve injury model of neuropathic pain. International journal of molecular sciences 21 (9). https://doi.org/10.3390/ijms21093396
Baxter LR Jr, Schwartz JM, Phelps ME, Mazziotta JC, Guze BH, Selin CE, Gerner RH, Sumida RM (1989) Reduction of prefrontal cortex glucose metabolism common to three types of depression. Arch Gen Psychiatry 46(3):243–250. https://doi.org/10.1001/archpsyc.1989.01810030049007
Hyder F, Patel AB, Gjedde A, Rothman DL, Behar KL, Shulman RG (2006) Neuronal-glial glucose oxidation and glutamatergic-GABAergic function. J Cereb Blood Flow Metab 26(7):865–877. https://doi.org/10.1038/sj.jcbfm.9600263
Patel AB, de Graaf RA, Mason GF, Rothman DL, Shulman RG, Behar KL (2005) The contribution of GABA to glutamate/glutamine cycling and energy metabolism in the rat cortex in vivo. Proc Natl Acad Sci U S A 102(15):5588–5593. https://doi.org/10.1073/pnas.0501703102
Giordano C, Cristino L, Luongo L, Siniscalco D, Petrosino S, Piscitelli F, Marabese I, Gatta L et al (2012) TRPV1-dependent and -independent alterations in the limbic cortex of neuropathic mice: impact on glial caspases and pain perception. Cereb Cortex 22(11):2495–2518. https://doi.org/10.1093/cercor/bhr328
Guida F, Luongo L, Marmo F, Romano R, Iannotta M, Napolitano F, Belardo C, Marabese I et al (2015) Palmitoylethanolamide reduces pain-related behaviors and restores glutamatergic synapses homeostasis in the medial prefrontal cortex of neuropathic mice. Mol Brain 8:47. https://doi.org/10.1186/s13041-015-0139-5
Spear LP (2000) The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev 24(4):417–463. https://doi.org/10.1016/s0149-7634(00)00014-2
Decosterd I, Woolf CJ (2000) Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain 87(2):149–158. https://doi.org/10.1016/s0304-3959(00)00276-1
Zhu J, Zhang Z, Jia J, Wang L, Yang Q, Wang Y, Chen C (2020) Sevoflurane induces learning and memory impairment in young mice through a reduction in neuronal glucose transporter 3. Cell Mol Neurobiol 40(6):879–895. https://doi.org/10.1007/s10571-019-00779-0
Kaidanovich-Beilin O, Lipina T, Vukobradovic I, Roder J, Woodgett JR (2011) Assessment of social interaction behaviors. J Vis Exp (48). https://doi.org/10.3791/2473
van der Kooij MA, Jene T, Treccani G, Miederer I, Hasch A, Voelxen N, Walenta S, Müller MB (2018) Chronic social stress-induced hyperglycemia in mice couples individual stress susceptibility to impaired spatial memory. Proc Natl Acad Sci U S A 115 (43):E10187-E10196. https://doi.org/10.1073/pnas.1804412115 %/ Copyright © 2018 the Author(s). Published by PNAS
Wu L, Niu Z, Hu X, Liu H, Li S, Chen L, Zheng D, Liu Z et al (2020) Regional cerebral metabolic levels and turnover in awake rats after acute or chronic spinal cord injury. FASEB J 34(8):10547–10559. https://doi.org/10.1096/fj.202000447R
Guo M, Fang Y, Zhu J, Chen C, Zhang Z, Tian X, Xiang H, Manyande A et al (2021) Investigation of metabolic kinetics in different brain regions of awake rats using the [(1)H-(13)C]-NMR technique. J Pharm Biomed Anal 204:114240. https://doi.org/10.1016/j.jpba.2021.114240%/Copyright©2021ElsevierB.V.Allrightsreserved
Vinall J, Pavlova M, Asmundson GJ, Rasic N, Noel M (2016) Mental health comorbidities in pediatric chronic pain: a narrative review of epidemiology, models, neurobiological mechanisms and treatment. Children (Basel, Switzerland) 3 (4). https://doi.org/10.3390/children3040040
Shelby GD, Shirkey KC, Sherman AL, Beck JE, Haman K, Shears AR, Horst SN, Smith CA et al (2013) Functional abdominal pain in childhood and long-term vulnerability to anxiety disorders. J Pediatrics. 132(3):475–482. https://doi.org/10.1542/peds.2012-2191
Maes M, Van den Noortgate W, Fustolo-Gunnink SF, Rassart J, Luyckx K, Goossens L (2017) Loneliness in children and adolescents with chronic physical conditions: a meta-analysis. J Pediatr Psychol 42(6):622–635. https://doi.org/10.1093/jpepsy/jsx046
Noel M, Wilson AC, Holley AL, Durkin L, Patton M, Palermo TM (2016) Posttraumatic stress disorder symptoms in youth with vs without chronic pain. Pain 157(10):2277–2284. https://doi.org/10.1097/j.pain.0000000000000642
Evans S, Djilas V, Seidman LC, Zeltzer LK, Tsao JCI (2017) Sleep quality, affect, pain, and disability in children with chronic pain: is affect a mediator or moderator? J Pain 18(9):1087–1095. https://doi.org/10.1016/j.jpain.2017.04.007
Humo M, Lu H, Yalcin I (2019) The molecular neurobiology of chronic pain-induced depression. Cell Tissue Res 377(1):21–43. https://doi.org/10.1007/s00441-019-03003-z
Ab I, Williams LM, Antees C, Grieve SM, Foster SL, Gomes L, Korgaonkar MS (2018) Cognitive ability is associated with changes in the functional organization of the cognitive control brain network. Hum Brain Mapp 39(12):5028–5038. https://doi.org/10.1002/hbm.24342
Fitzgerald M, McKelvey R (2016) Nerve injury and neuropathic pain - a question of age. Exp Neurol 275(Pt 2):296–302. https://doi.org/10.1016/j.expneurol.2015.07.013
Vega-Avelaira D, McKelvey R, Hathway G, Fitzgerald M (2012) The emergence of adolescent onset pain hypersensitivity following neonatal nerve injury. Mol Pain 8:30. https://doi.org/10.1186/1744-8069-8-30
Jais A, Solas M, Backes H, Chaurasia B, Kleinridders A, Theurich S, Mauer J, Steculorum SM, et al (2016) Myeloid-cell-derived VEGF maintains brain glucose uptake and limits cognitive impairment in obesity. Cell 165 (4):882-895. https://doi.org/10.1016/j.cell.2016.03.033 %/ Copyright © 2016 Elsevier Inc. All rights reserved.
Krell-Roesch J, Syrjanen JA, Vassilaki M, Lowe VJ, Vemuri P, Mielke MM, Machulda MM, Stokin GB et al (2021) Brain regional glucose metabolism, neuropsychiatric symptoms, and the risk of incident mild cognitive impairment: the Mayo Clinic study of aging. Am J Geriatr Psychiatry 29(2):179–191. https://doi.org/10.1016/j.jagp.2020.06.006
Cunnane S, Nugent S, Roy M, Courchesne-Loyer A, Croteau E, Tremblay S, Castellano A, Pifferi F et al (2011) Brain fuel metabolism, aging, and Alzheimer’s disease. Nutrition (Burbank, Los Angeles County, Calif) 27(1):3–20. https://doi.org/10.1016/j.nut.2010.07.021
Chen Z, Zhong C (2013) Decoding Alzheimer's disease from perturbed cerebral glucose metabolism: implications for diagnostic and therapeutic strategies. Prog Neurobiol 108:21-43. doi:https://doi.org/10.1016/j.pneurobio.2013.06.004 %/ Copyright © 2013 Elsevier Ltd. All rights reserved
Su J, Huang Q, Ren S, Xie F, Zhai Y, Guan Y, Liu J, Hua F (2019) Altered brain glucose metabolism assessed by (18)F-FDG PET imaging is associated with the cognitive impairment of CADASIL. Neuroscience 417:35-44. doi:https://doi.org/10.1016/j.neuroscience.2019.07.048 %/ Copyright © 2019 IBRO. All rights reserved
Magistretti PJ, Allaman I (2015) A cellular perspective on brain energy metabolism and functional imaging. Neuron 86(4):883–901. https://doi.org/10.1016/j.neuron.2015.03.035
Cunnane SC, Trushina E, Morland C, Prigione A, Casadesus G, Andrews ZB, Beal MF, Bergersen LH et al (2020) Brain energy rescue: an emerging therapeutic concept for neurodegenerative disorders of ageing. Nat Rev Drug Discov 19(9):609–633. https://doi.org/10.1038/s41573-020-0072-x
Mao J, Mayer DJ, Price DD (1993) Patterns of increased brain activity indicative of pain in a rat model of peripheral mononeuropathy. J Neurosci 13(6):2689–2702. https://doi.org/10.1523/jneurosci.13-06-02689.1993
Vannucci SJ, Seaman LB, Brucklacher RM, Vannucci RC (1994) Glucose transport in developing rat brain: glucose transporter proteins, rate constants and cerebral glucose utilization. Mol Cell Biochem 140(2):177–184. https://doi.org/10.1007/bf00926756
Pearson-Leary J, McNay EC (2016) Novel roles for the insulin-regulated glucose transporter-4 in hippocampally dependent memory. J Neurosci 36 (47):11851-11864. https://doi.org/10.1523/JNEUROSCI.1700-16.2016 %/ Copyright © 2016 the authors 0270-6474/16/3611851-14$15.00/0
Ye T, Meng X, Zhai Y, Xie W, Wang R, Sun G, Sun X (2018) Gastrodin ameliorates cognitive dysfunction in diabetes rat model via the suppression of endoplASMIC RETICULUM STRESS and NLRP3 inflammasome activation. Front Pharmacol 9:1346. https://doi.org/10.3389/fphar.2018.01346
Pang R, Wang X, Pei F, Zhang W, Shen J, Gao X, Chang C (2019) Regular exercise enhances cognitive function and intracephalic GLUT expression in Alzheimer’s disease model mice. J Alzheimers Dis 72(1):83–96. https://doi.org/10.3233/jad-190328
Peek AL, Rebbeck T, Puts NA, Watson J, Aguila MR, Leaver AM (2020) Brain GABA and glutamate levels across pain conditions: a systematic literature review and meta-analysis of 1H-MRS studies using the MRS-Q quality assessment tool. Neuroimage 210:116532. doi:https://doi.org/10.1016/j.neuroimage.2020.116532 %/ Copyright © 2020 The Authors. Published by Elsevier Inc. All rights reserved
Ji G, Sun H, Fu Y, Li Z, Pais-Vieira M, Galhardo V, Neugebauer V (2010) Cognitive impairment in pain through amygdala-driven prefrontal cortical deactivation. J Neurosci 30(15):5451–5464. https://doi.org/10.1523/jneurosci.0225-10.2010
Ji G, Neugebauer V (2011) Pain-related deactivation of medial prefrontal cortical neurons involves mGluR1 and GABA(A) receptors. J Neurophysiol 106(5):2642–2652. https://doi.org/10.1152/jn.00461.2011
Xiong B, Zhang W, Zhang L, Huang X, Zhou W, Zou Q, Manyande A, Wang J et al (2020) Hippocampal glutamatergic synapses impairment mediated novel-object recognition dysfunction in rats with neuropathic pain. Pain 161(8):1824–1836. https://doi.org/10.1097/j.pain.0000000000001878
Barros LF (2013) Metabolic signaling by lactate in the brain. Trends Neurosci 36(7):396–404. https://doi.org/10.1016/j.tins.2013.04.002
Magistretti PJ, Allaman I (2018) Lactate in the brain: from metabolic end-product to signalling molecule. Nat Rev Neurosci 19(4):235–249. https://doi.org/10.1038/nrn.2018.19
Proia P, Di Liegro CM, Schiera G, Fricano A, Di Liegro I (2016) Lactate as a metabolite and a regulator in the central nervous system. International journal of molecular sciences 17 (9). https://doi.org/10.3390/ijms17091450
Nilsson L, Siesjo BK (1975) The effect of phenobarbitone anaesthesia on blood flow and oxygen consumption in the rat brain. Acta Anaesthesiol Scand Suppl 57:18–24. https://doi.org/10.1111/j.1399-6576.1975.tb05408.x
Liu T, Li Z, He J, Yang N, Han D, Li Y, Tian X, Liu H et al (2020) Regional metabolic patterns of abnormal postoperative behavioral performance in aged mice assessed by 1H-NMR dynamic mapping method. Neurosci Bull 36(1):25–38. https://doi.org/10.1007/s12264-019-00414-4
Acknowledgements
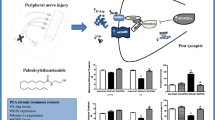
We wish to thank Yingying Chen, Department of Anaesthesiology, Zhongnan Hospital, Wuhan University, for her help in cartoon illustrating.
Funding
This work was supported by the National Natural Science Foundation of China (NO. 81771160, 31771193 and 81901109).
Author information
Authors and Affiliations
Contributions
Yuanyuan Fang, Chang Chen, Jie Wang, and Zongze Zhang designed the study; Yuanyuan Fang, Qi Zhong, Lirong Wang, Zhu Gui, and Jinpiao Zhu performed the experiments; Yuanyuan Fang, Chang Chen, Jie Wang, and Fuqiang Xu contributed to the data. Yuanyuan Fang, Chang Chen, Jie Wang, Zongze Zhang, and Anne Manyande wrote the manuscript. The content of this manuscript has been reviewed, read, and agreed upon by all the designated authors.
Corresponding authors
Ethics declarations
Ethics Approval
All animal procedures were carried out in accordance with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals and were approved by the Animal Ethics Committee of Zhongnan Hospital of Wuhan University (Ethics approval number: ZN2021097).
Consent to Participate
Not applicable.
Consent for Publication
All the authors have read the manuscript and agreed for its publication.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fang, Y., Chen, C., Zhong, Q. et al. Influence of Cerebral Glucose Metabolism by Chronic Pain–Mediated Cognitive Impairment in Adolescent Rats. Mol Neurobiol 59, 3635–3648 (2022). https://doi.org/10.1007/s12035-022-02816-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-022-02816-4